With new research and advancements in technology becoming available each and every day, we strive to offer the newest, safest and most efficient instruments and procedures to our patients. Here are a few recent developments in glaucoma treatment that we offer to patients in the greater Minneapolis, Minnesota area.
The XEN Gel Stent is a procedure that is most analogous to a Trabeculectomy/ExPress Shunt. It is, however, much more refined and less invasive than Trabeculectomy. The XEN Gel Stent is ideal for patients with more advanced glaucoma and can be used as a standalone procedure independent of cataract surgery.
Like Trabeculectomy, the XEN Gel Stent forms a filtration bleb, intended to significantly reduce and sustain the patient’s intraocular eye pressure. Although this treatment does have a slower visual recovery than the iStent procedure, it likely lowers pressure more effectively (although there are no comparative studies).
The pliability and softness of the gel stent allow it to conform to the tissue within the eye and minimize complications related to synthetic materials. The XEN Gel Stent has been approved in Europe and in Canada for many years and has been used extensively in these regions of the world.
The MicroPulse laser treatment is a non-invasive therapy that delivers repetitive, low-energy laser ‘micropulse’ above and below the iris, with the goal of lowering the patient’s eye pressure. This treatment is performed within the OR so anesthesia can be administered during the procedure.
Some perks to using this laser system are the procedure is repeatable and the patient’s downtime after is minimal.
Most patients do not experience pain after the therapy, but may be asked to wear a patch for the remainder of the day.
GATT is a modified Trabeculectomy – a procedure commonly performed at Minnesota Eye Consultants. A Trabeculectomy creates a new drainage system from the eye’s natural tissue for the aqueous humor (the transparent fluid within the eye that can cause high pressures) to exit, while the GATT procedure aims at enhancing the patient’s own natural drainage system.
Through a small incision, a tiny catheter is placed and rotated around the Schlemm’s canal 360 degrees. This minimally invasive surgery treats patients with open-angle and is performed within our Ambulatory Surgery Centers.
The Kahook Dual Blade (KDB) is an ophthalmic blade that makes parallel incisions in the trabecular meshwork and inner wall of the Canal of Schlemm. Incisions created with this device precisely remove tissue from the meshwork and as a result, help reduce intraocular pressure behind the eye.
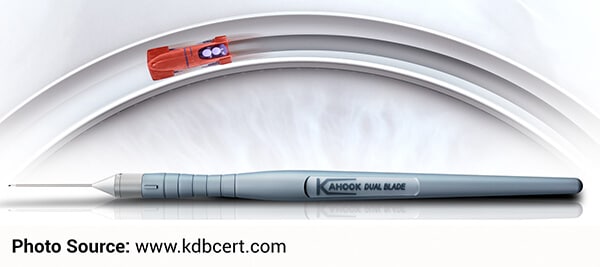
On the day of the procedure, the surgeon will determine whether or not this innovative surgical tool will be beneficial during the surgery. Currently, this device is available at our Ambulatory Surgery Centers.
Sources: medgadget.com, glaucomaassociates.com, www.eyeworld.org, www.kdbcert.com, iridex.com, and aquesys.com
The CyPass micro-stent is an incredibly small device used to create a new channel for fluid to flow out of the eye, ultimately reducing eye pressure for our patients. This implant is also used during Cataract surgery. The FDA approved CyPass in 2016 based on favorable two-year safety and effectiveness data.
Since approval, CyPass has been implanted in tens of thousands of eyes worldwide and has become a popular surgical treatment for the management of the disease.
While the Cypass device was approved based on two-year data, the FDA required a follow up “post-approval” study to provide additional safety data. The 5-year safety data are now available, and the results were recently released.
While the effectiveness results remain favorable, some patients had an accelerated loss of corneal endothelial cells, the very delicate cells that line the inside of the cornea. Some patients were found to be at higher risk than others, and the risk was closely correlated to the position of the CyPass within the eye. Fortunately, no eye has had any serious consequence so far, however, all eyes that have received CyPass will require ongoing monitoring.
Based on this new information, Alcon has decided to voluntarily withdraw CyPass from the global market while it investigates the significance of these findings.
Minnesota Eye Consultants is proud to offer patients convenient access to eye care across the Twin Cities. We have 5 locations, each with an onsite ambulatory surgery center (ASC).